Triple-negative breast cancer (TNBC) is one of the most challenging breast cancer subtypes to treat. Unlike hormone-positive or HER2-positive tumours, TNBC does not respond to the targeted therapies that have transformed outcomes for many other patients. For UK women diagnosed with this subtype, the path through treatment can feel uncertain, and the search for additional options is entirely understandable.
In recent years, growing numbers of patients from across the UK and Europe have begun exploring immunotherapy protocols offered at specialised German laboratories. Central to this interest is Dendritic cell therapy for triple-negative breast cancer. This approach uses a patient’s own immune cells to help the body better recognise and respond to the tumour. This guide explains what dendritic cell therapy is, how it is delivered in Germany, what current evidence shows, and how UK patients can access it safely and realistically.
Understanding Triple-Negative Breast Cancer: Why Fewer Options Exist
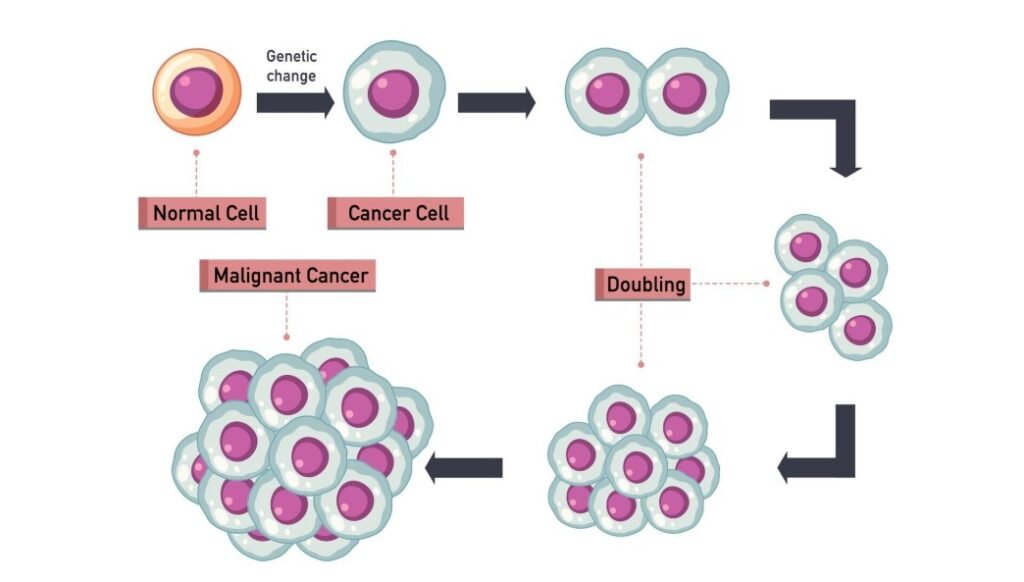
Triple-negative breast cancer accounts for approximately 10 to 15 percent of all breast cancer diagnoses. It is defined by the absence of three key markers: oestrogen receptors, progesterone receptors, and the HER2 protein. This combination means that hormonal therapies such as tamoxifen and targeted drugs such as trastuzumab (Herceptin) have no effect. Standard treatment relies primarily on chemotherapy, surgery, and, in some cases, radiotherapy.
According to Cancer Research UK, TNBC is more common in younger women, those of Black African or Caribbean heritage, and individuals carrying BRCA1 gene mutations. It tends to grow and spread more rapidly than other breast cancer subtypes, and the risk of recurrence is highest within the first three to five years following initial treatment.
The absence of hormone receptors and HER2 also affects how the immune system interacts with the tumour. Without the distinctive surface markers that other breast cancers carry, TNBC cells can be harder for the immune system to detect, which is one of the reasons researchers and oncologists have shown increasing interest in immune-based approaches for this subtype.
What Is Dendritic Cell Therapy and How Does It Work
Dendritic cells are specialised immune cells that act as messengers between the innate and adaptive immune systems. Their primary function is to identify foreign or abnormal material, process it, and present it to T-cells in a way that triggers an immune response. In cancer, this process is often disrupted: tumour cells can evade detection, suppress immune signalling, or fail to produce the signals that dendritic cells need to mount an effective response.
Dendritic cell therapy, sometimes referred to as DCT or a dendritic cell vaccine, works by enhancing this natural process outside the body. The patient’s own blood is drawn, and the precursor cells that can develop into dendritic cells are isolated. These are grown in laboratory conditions until they mature, then exposed to tumour-specific material taken from the patient’s own biopsy or surgical sample. The trained dendritic cells are then returned to the patient as a series of injections.
The goal is to give the immune system clearer, more structured instructions about what the tumour looks like, so that T-cells can mount a more organised and targeted attack. For dendritic cell therapy for triple-negative breast cancer, this approach holds particular biological relevance because the tumour provides few natural immune signals on its own.
Key Steps in the Dendritic Cell Protocol
- Blood draw: A sample of the patient’s blood is taken at the clinic.
- Cell isolation: Monocytes (precursor cells) are separated from the blood sample
- Maturation: Cells are cultured over several days under controlled laboratory conditions until they develop into mature dendritic cells
- Antigen loading: The mature dendritic cells are exposed to tumour-specific antigens, typically derived from the patient’s own biopsy material
- Quality control: The prepared vaccine is tested for safety, sterility, and viability
- Administration: The vaccine is delivered as a series of subcutaneous or intradermal injections over a defined schedule
- Follow-up: Immune response markers and imaging are monitored over subsequent weeks and months
Why UK Patients with TNBC Are Looking Beyond Standard Treatment
For most UK patients, TNBC is treated within the NHS using established protocols: chemotherapy before or after surgery, sometimes combined with newer immunotherapy agents such as pembrolizumab (Keytruda), which was approved by NICE for certain TNBC patients in 2022. These are important advances, but they do not work for every patient, and not every TNBC case meets the criteria for pembrolizumab eligibility.
Patients who have completed standard treatment and are in remission sometimes seek additional ways to support their immune system during this higher-risk period. Others who experience progression or recurrence may be exploring options alongside or after further systemic therapy. In both situations, the desire to take a proactive role in immune support is a consistent theme.
Interest in dendritic cell protocols also reflects a broader shift in how patients think about cancer care. Rather than viewing treatment as a series of passive interventions, more people are looking for approaches that actively engage the body’s own defences. Dendritic cell therapy appeals in this context because it is derived entirely from the patient’s own cells and does not introduce external chemical agents.
Germany as a Centre for Advanced Immunotherapy: What UK Patients Should Know
Germany has developed one of the most established frameworks for cell-based cancer immunotherapy in Europe. Several specialised laboratories operate under strict manufacturing standards aligned with Good Manufacturing Practice (GMP) guidelines, which govern the quality and safety of biologically derived medical products. This regulatory infrastructure is one reason patients from the UK, the United States, and across the Middle East have sought treatment there.
For UK patients considering treatment abroad, identifying the best cancer hospitals for international patients involves looking beyond promotional materials. The most reputable German centres are transparent about the evidence base for their protocols, conduct thorough pre-treatment evaluations, and maintain open communication with the patient’s NHS oncologist at home. They do not present dendritic cell therapy as a cure, but as a potential immune support strategy that may complement ongoing or completed conventional treatment.
UK patients should also be aware that the NHS does not fund treatment abroad in most cases. That prior authorisation under the UK’s Global Health Insurance arrangements is unlikely to cover experimental or investigational protocols. Costs should be fully understood before any commitment is made.
What to Look for When Evaluating a German Clinic
- GMP-certified laboratory with documented dendritic cell manufacturing processes
- Willingness to review your full medical history, imaging, and pathology reports before accepting your case
- Clear communication about what outcomes are realistic and what cannot be promised
- Experience specifically with breast cancer cases, including TNBC
- An established process for liaising with your UK-based oncologist
- Transparent pricing and a written treatment plan before any payment is requested
- Access to verified patient reviews or testimonials from previous international patients
Inside German Dendritic Cell Laboratories: What the Process Involves
Laboratory Standards and Manufacturing
The quality of the dendritic cell product depends entirely on the precision of the laboratory process. In Germany, laboratories that produce cell-based therapies for clinical use are expected to operate under GMP conditions, which regulate everything from staff training and equipment calibration to sterility testing and documentation. This is comparable to the standards applied to pharmaceutical manufacturing.
The maturation process for dendritic cells typically involves specific cytokine cocktails, which are combinations of signalling proteins that guide the precursor cells through their development into functional antigen-presenting cells. The choice of cytokines, the duration of culture, and the type of tumour antigens used can all influence the quality and immunological potency of the final product.
Tumour Antigen Sourcing
For the dendritic cells to present relevant tumour information to the immune system, they need to be loaded with material that reflects the patient’s specific cancer. Some German centres use lysate from the patient’s own surgically removed tumour, which contains a broad mixture of tumour proteins. Others use defined tumour-associated peptides, which are shorter, more specific antigen fragments. Each approach has its own advantages in terms of breadth versus specificity of immune activation.
Injection Schedules and Follow-Up
The vaccination schedule varies between centres, but most protocols involve an initial series of injections delivered over several weeks, followed by booster injections at defined intervals. Some clinics also incorporate adjuvants, additional immune-stimulating agents, alongside the dendritic cell injections to enhance the immune response. Follow-up involves periodic blood tests to measure immune cell activity and imaging to assess disease status.
What the Evidence Currently Shows for Dendritic Cell Therapy in Breast Cancer
Clinical data on dendritic cell therapy in breast cancer are still maturing. This is an important point for UK patients to understand clearly: this is not a standard-of-care treatment, and the evidence base is not yet at the level required for NHS approval or NICE guidance. What exists, however, is a growing body of early-phase clinical trial data and observational research that has produced some consistently interesting findings.
Early research has demonstrated immune activation in approximately 40 to 60 percent of patients following dendritic cell vaccination. In some cases, increased T-cell activity directed against tumour-associated antigens has been detected in blood tests. Some studies have observed periods of disease stabilisation following treatment, particularly in patients who have already reduced their tumour burden through surgery or chemotherapy.
Research published in Nature Communications has highlighted that dendritic cell-based immunotherapy has shown the most consistent immune responses when administered after debulking of the primary tumour, suggesting that timing relative to conventional treatment may influence outcomes. Separately, early data from triple-negative breast cancer-specific trials, including work from institutions in Germany and the Netherlands, have demonstrated that the tumour microenvironment in TNBC may actually be more amenable to immune activation than previously thought, partly because TNBC tumours tend to have higher rates of tumour-infiltrating lymphocytes.
What the evidence does not yet show is a clear, statistically significant improvement in overall survival attributable to dendritic cell therapy alone in a large randomised controlled trial for TNBC. UK patients considering this treatment should hold both parts of this picture: genuine immunological signals of activity on one hand, and the absence of definitive survival data on the other.
Questions UK Patients Should Ask Before Travelling for Dendritic Cell Therapy
For UK women evaluating treatment abroad, the quality of the consultation process is itself an important indicator of how a clinic operates. The following questions are worth raising directly with any centre being considered:
- What tumour type and stage is this protocol most commonly used for, and do you have documented outcomes for TNBC specifically?
- Is your laboratory GMP-certified, and can you provide documentation?
- How do you source and process the tumour antigens used to train the dendritic cells?
- What does your injection schedule look like, and how many visits to Germany will be required?
- What immune monitoring is included, and how will you assess whether the therapy is producing a response?
- Will you communicate directly with my NHS oncologist, and can you share treatment summaries with my UK team?
- What is the total cost, including laboratory preparation, injections, follow-up, and any additional supportive treatments?
- What happens if I need medical support between visits while I am back in the UK?
How UK Patients Can Access Dendritic Cell Therapy in Germany
The process of accessing dendritic cell therapy in Germany follows a structured sequence. Understanding what to expect at each stage helps reduce uncertainty and allows patients to plan realistically.
Step 1: Preparing Your Medical Records
German clinics require a comprehensive set of documents before they can assess suitability. These typically include pathology reports confirming the TNBC diagnosis and receptor status, recent imaging (CT, MRI, or PET-CT), a summary of treatments received to date, including chemotherapy regimens and dates, surgical notes, and any existing blood test or immune marker results. Documents should ideally be translated into English if they are not already, although most German centres accepting international patients have staff who can manage this.
Step 2: Initial Case Review
Once documents are submitted, a specialist at the clinic reviews the case to assess whether the patient is a suitable candidate for the protocol. This review considers the current disease status, the interval since last treatment, general health and performance status, and any factors that might affect the timing or safety of cell preparation. Not all patients will be considered suitable, and reputable clinics will explain their reasoning clearly.
Step 3: Consultation
If the initial review is positive, a consultation is arranged, either in person in Germany or via video call. The treating physician explains the specific protocol to be used, the realistic range of possible outcomes, the injection schedule, and what monitoring will be involved. This is the appropriate stage to ask all the questions listed in the previous section.
Step 4: Treatment Planning and Travel
Most patients make two visits to Germany: one for the blood draw, and a second for the injection series once the vaccine has been prepared, which typically takes one to two weeks. Some centres allow the blood draw to take place locally and have the sample transported to the laboratory under specific conditions, though this varies by clinic and protocol. Follow-up appointments can often be handled remotely, with blood tests and imaging conducted through the patient’s NHS GP or oncologist in the UK.
Step 5: Ongoing Monitoring
After the injection series is complete, monitoring continues through periodic immune blood tests and imaging. Most German clinics ask patients to share results from these tests, which can be arranged with their UK GP. Communication between the German treating team and the patient’s NHS oncologist is strongly advisable throughout, both to ensure safety and to allow the NHS team to have a complete picture of all treatments being received.
Using Healthcare Platforms to Compare Clinics and Specialists
One of the practical challenges of seeking treatment abroad is the difficulty of comparing clinics fairly and efficiently. Specialist healthcare platforms have emerged to address this, providing structured profiles of international centres, including their focus areas, laboratory capabilities, and documented experience with specific cancer types.
Platforms of this kind allow patients to review clinic protocols side by side, access verified feedback from previous patients, and share documents with multiple centres simultaneously. They also facilitate initial consultations and can help patients understand the differences between how individual centres approach the dendritic cell manufacturing process.
When using any platform to research treatment options, UK patients should apply the same critical scrutiny they would to any medical decision. Look for transparent information about clinic accreditation, honest communication about what can and cannot be achieved, and clear pricing before any commitment is made. A reputable platform will not guarantee outcomes and will make it straightforward to obtain a second opinion.
Coordinating Dendritic Cell Therapy with Your NHS Care Team
One of the most important steps UK patients can take is to keep their NHS oncologist fully informed about any treatment being pursued abroad. This is not simply a matter of courtesy. It is essential for safety. Some immune-stimulating treatments can interact with chemotherapy or other systemic therapies, and your NHS team needs a complete picture of all interventions you are receiving.
UK patients are also encouraged to raise the subject of immunotherapy options, including emerging protocols, directly with their oncologist. While NHS oncologists cannot typically refer patients to German clinics for investigational treatments, they can help contextualise the evidence, flag any potential concerns about timing or drug interactions, and, in some cases, support monitoring through NHS blood tests or imaging.
The Breast Cancer Now charity and Macmillan Cancer Support both offer dedicated support services for women with TNBC, including nurse helplines, peer support groups, and access to clinical trial information. These resources can be invaluable for patients navigating complex treatment decisions.
Clinical Trials for TNBC Immunotherapy Available to UK Patients
Before travelling abroad for investigational treatment, it is worth exploring whether relevant clinical trials are currently recruiting in the UK. The NIHR Clinical Trials Gateway and the Cancer Research UK Clinical Trials Database both allow patients to search for studies by cancer type.
Several UK institutions, including The Royal Marsden NHS Foundation Trust and University College London Hospitals, are active in breast cancer immunotherapy research. Participating in a clinical trial not only provides access to cutting-edge treatments under close medical supervision, but also contributes to the evidence base that will ultimately determine whether approaches like dendritic cell therapy become standard care.
Clinical trials are not available to all patients, and eligibility criteria can be strict. However, even an exploratory conversation with a trial coordinator can provide useful information about where the research landscape currently stands.
Final Thoughts
For UK women living with triple-negative breast cancer, the desire to explore every available option is entirely reasonable. Dendritic cell therapy represents a genuinely interesting area of immunological research with a plausible biological rationale for TNBC specifically. The clinical signals so far are encouraging enough to justify continued investigation, and the best German laboratories operate to high manufacturing standards with a realistic, evidence-informed approach to patient care.
At the same time, this is not an established treatment with proven survival benefits in large randomised trials. UK patients should approach it as a complementary strategy, one that may support immune function alongside or after conventional treatment, rather than as an alternative to it. The decision to travel abroad for any medical treatment deserves the same careful evaluation as any major health decision: independent research, professional medical advice, transparent cost information, and honest conversations with your NHS team.
With realistic expectations, thorough preparation, and the right clinical partners, some patients do find value in pursuing these protocols. The goal is always to make a decision that feels medically sound, personally informed, and free from pressure.
Medical Disclaimer
The information in this article is provided for general educational and informational purposes only. It does not constitute medical advice and should not be used as a substitute for professional guidance from a qualified healthcare provider, including your GP, oncologist, or cancer care team. Dendritic cell therapy is not a standard NHS treatment for triple-negative breast cancer and is not approved by NICE for this indication. Clinical outcomes vary significantly between individuals and cannot be predicted or guaranteed. Patients are strongly encouraged to discuss all treatment options, including any therapies pursued abroad, with their NHS oncologist before proceeding. The author and publisher accept no responsibility for decisions made based on this article.
References
- National Cancer Institute (NCI). Immunotherapy for Cancer. Available at: https://www.cancer.gov/about-cancer/treatment/types/immunotherapy
- Cancer Research UK. Triple Negative Breast Cancer. Available at: https://www.cancerresearchuk.org/about-cancer/breast-cancer/stages-types-grades/types/triple-negative-breast-cancer
- Breast Cancer Now. Triple Negative Breast Cancer Information and Support. Available at: https://breastcancernow.org/information-support/have-i-got-breast-cancer/triple-negative-breast-cancer
- Macmillan Cancer Support. Breast Cancer Information. Available at: https://www.macmillan.org.uk/cancer-information-and-support/breast-cancer
- National Institute for Health and Care Research (NIHR). Find a Clinical Trial. Available at: https://www.nihr.ac.uk/patients-and-public/i-want-to-take-part-in-research
- Cancer Research UK. Find a Clinical Trial Database. Available at: https://www.cancerresearchuk.org/about-cancer/find-a-clinical-trial
- World Health Organization (WHO). Cancer Overview. Available at: https://www.who.int/health-topics/cancer
- Bol KF, Schreibelt G, de Vries IJM. (2022). Dendritic cell-based cancer immunotherapy: Advances in clinical trials and manufacturing. Nature Communications.
- Saxena M, Bhardwaj N, Reizis B. (2021). Dendritic-cell immunotherapy: From ex vivo loading to in vivo targeting. Nature Reviews Immunology.
- Sabado RL, Bhardwaj N. (2020). Dendritic cell immunotherapy for cancer: Current status and future directions. Seminars in Immunology. Available via: https://pubmed.ncbi.nlm.nih.gov/