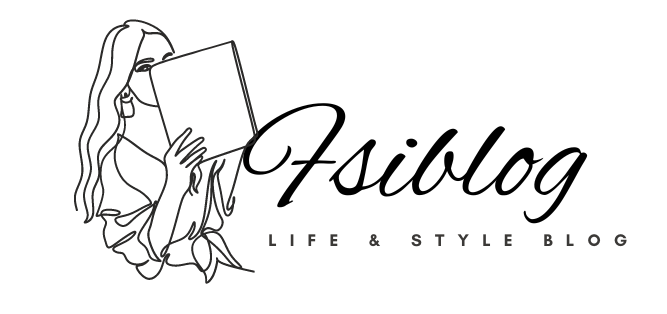A digital content factory in pharma gives healthcare teams a practical way to support transparency. It turns scattered files, email approvals, and local edits into one controlled system for content authoring and approval, reuse, localization, and omnichannel activation. When every claim, reference, disclaimer, and audience rule lives in a structured flow, teams publish faster, track every change, and keep records ready for review.
Why a Digital Content Factory for Pharma Changes Content Operations
Many pharma content workflows still begin with a slide deck, a Word file, or a local spreadsheet. That sounds manageable at first. It breaks when the same message must appear on a website, in a rep email, in a patient leaflet, in a banner, and in a medical review packet.
A pharma content factory fixes that by treating content as structured components instead of isolated files. The model works well for pharma digital content operations because it separates content from layout. One approved claim can feed many channels. One updated safety line can move across markets without manual copy-paste in ten places.
This is where a digital content factory for pharma starts to outperform older pharmaceutical content workflows. It reduces rework, gives legal and regulatory teams better visibility, and supports faster release cycles without losing control.
| Old workflow | Factory workflow | Result |
| Content lives in channel files | Content lives in reusable modules | Fewer duplicate edits |
| Approval happens by email | Approval happens in tracked steps | Clear audit trail |
| Local teams rewrite assets | Local teams adapt approved blocks | Better consistency |
Digital Content Factory for Pharma: From Draft to Submission-Ready Content
A mature pharma content production workflow starts with claims, references, audience rules, metadata, and channel logic, not with final layout. That shift changes how teams build submission-ready content.
Content authoring and approval
Writers create modular blocks: headline, body copy, safety statement, indication, reference note, CTA, and local disclaimer. Medical, legal, and regulatory reviewers approve each block at the right level. This turns content authoring and approval into a repeatable operating model rather than a chain of ad hoc comments.
Regulatory content management
Strong regulatory content management keeps approved text linked to evidence, version history, market rules, and expiration dates. When a label update arrives, teams can find every affected block instead of searching old PDFs and inbox threads.
Approved content reuse
When teams build content with clear structure and tagging, approved content reuse lets one medical statement move from an HCP email into a detail aid, landing page, CRM sequence, and webinar follow-up.
Lifecycle content management
Because pharma materials keep changing across launch, uptake, safety updates, line extensions, and withdrawal, lifecycle content management keeps status, owners, review dates, and retirement rules visible at each stage.
Content compliance automation
Before reviewers spend time on an asset, content compliance automation can flag missing references, expired claims, wrong audience labels, market restrictions, and off-template safety copy through rule-based checks.
Healthcare Transparency Needs Structured Content, Not More Content
Healthcare transparency is often treated as a publishing issue. In practice, it is a content architecture issue. If the same product message appears in five places with three wording variations, trust starts to erode. The problem is larger in pharma content operations, where every update can affect multiple audiences and multiple countries.
Take a common scenario. A brand team updates one safety statement. Without structure, the team must touch a website page, an email journey, a PDF detail aid, a webinar deck, and a local market adaptation. One missed asset creates inconsistency. In a factory model, the safety line sits in one approved module, linked to all dependent assets. Update once, publish many.
That is the practical value of a digital content factory in pharma. It gives teams one source for claims, references, and safety language, while still allowing controlled channel variation.
Pharma Digital Content Operations Need a Common Taxonomy
Without a shared taxonomy, even good content becomes hard to find. Teams label files by campaign name, product nickname, or market habit. Six months later, no one knows which block is current.
A working content tagging taxonomy usually includes:
- Product and indication.
- Audience type.
- Market and language.
- Claim type.
- Medical or promotional status.
- Approval status.
- Expiry or review date.
- Channel eligibility.
That structure supports content standardization without flattening all content into the same tone. It also improves search, localization, review routing, and omnichannel reuse.
Here is a compact example:
| Content block | Metadata tags | Use case |
| Efficacy claim | HCP, US, approved, reference-linked | Website and email |
| Safety text | Global, required, current label | All channels |
| CTA module | Local, channel-specific, approved | CRM and landing page |
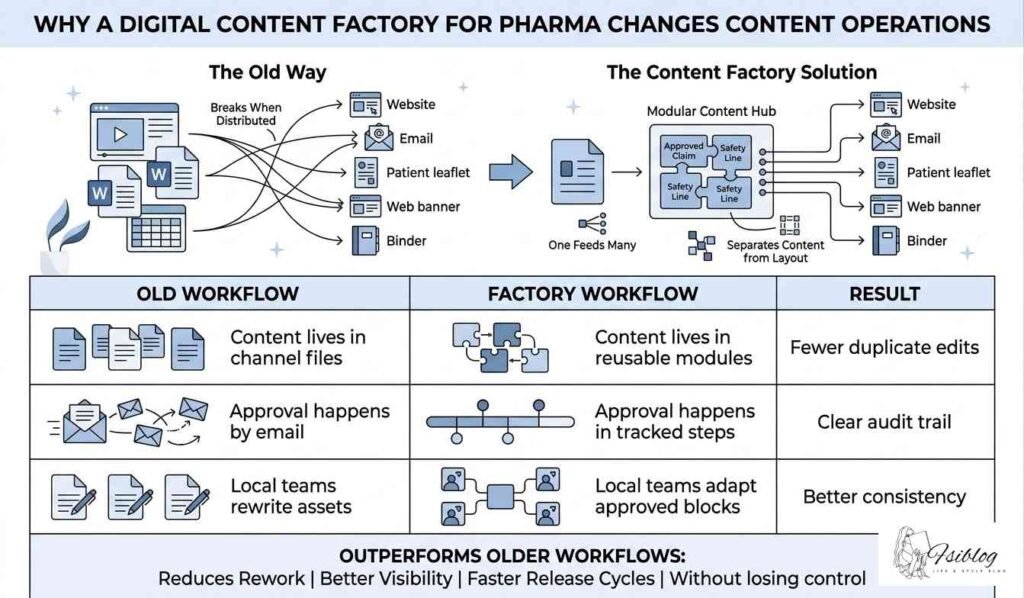
Digital Content Factory for Pharma: A Working Model by Team
The operating model becomes clearer when each team owns a defined part of the process.
- Brand and medical teams define the message framework.
- Content teams write modular copy and map reusable blocks.
- Regulatory and legal reviewers approve at block level.
- Operations teams apply metadata and workflow rules.
- Local teams handle global-to-local content adaptation within allowed boundaries.
- Publishing teams trigger omnichannel activation across web, email, CRM, portals, and sales tools.
This is how pharma content supply chain thinking enters the picture. The phrase can sound operational, yet the idea is simple: content moves through a controlled production system with clear inputs, controls, outputs, and reuse points.
Teams searching for regulated content workflows pharma usually want four things at once: traceability, reuse, speed, and local control. A factory model gives them a way to balance all four.
Pharma Content Factory Economics: A Reuse Calculation
A content factory becomes easier to justify when the math is visible.
Assume a launch team needs:
- 8 core claims.
- 3 audience variants.
- 4 channels.
- 5 markets.
A file-based model may push the team toward 8 × 3 × 4 × 5 = 480 content instances to draft, review, and store.
Now assume the team builds those claims as approved modules with shared safety copy and market rules. If 60% of the content is reusable across channels and 50% of local variation is handled through metadata and approved local layers, the number of net unique build tasks drops sharply. The team still governs 480 outputs, but it no longer authors 480 separate pieces.
That is the hidden gain in a pharma content factory. The output count stays high. The authoring burden drops because reuse sits inside the system.
Healthcare Web Development Connects the Factory to the Front End
A content factory does not stop at storage and approval. It must connect to the places where content appears. That is where the web layer becomes part of the stack. The website, portal, or product page should consume approved modules, preserve metadata, and respect channel rules instead of relying on manual pasting from source documents.
This connection matters for three reasons:
- Web teams publish faster when approved content is already structured.
- Reviewers see the relationship between source modules and rendered pages.
- Updates reach live experiences with fewer manual handoffs.
When teams keep content operations and front-end delivery separate for too long, they create bottlenecks. Content gets approved in one system and rebuilt in another. That gap is where errors usually appear.
What Teams Miss When Building a Digital Content Factory for Pharma
Most problems do not come from the idea. They come from execution.
Common failure points include:
- Treating templates as the same thing as structured content.
- Skipping metadata design until late in the project.
- Approving full assets instead of reusable blocks.
- Giving local teams too little room to adapt content.
- Storing references outside the content object.
- Leaving retired claims in active search results.
- Building workflows around channels instead of message components.
A strong setup creates audit-ready content workflows from the start. That means every block has an owner, status, review path, evidence link, and retirement rule.
A Practical Checklist for a Digital Content Factory for Pharma
Before rollout, teams should be able to answer these questions:
- Where does approved source content live?
- Which metadata fields are mandatory?
- Can one approved block feed more than one channel?
- How are local deviations recorded?
- What triggers re-approval?
- How does the system mark expired content?
- Can reviewers trace rendered assets back to the approved source?
If those answers are unclear, the operating model still needs work.
Closing Remarks
The best version of a digital content factory in pharma is not a content warehouse. It is a controlled production model for regulated messaging. It supports reuse, faster review, clearer traceability, and better channel delivery. In practice, that leads to cleaner pharma content operations, more reliable regulatory content management, and content teams that spend less time rebuilding what they already approved.
Digital content factory in pharma works when content is modular, tagged, reviewable, and connected to the systems where it will actually appear. That is the point where healthcare transparency stops being a slogan and starts becoming an operating standard.